30-second summary
Composition of Battery
Batteries are made of an extensive range of materials resulting in different capabilities and behaviors in the functionality of the battery. The most common ones are lead, nickel, zinc, and lithium.
The chemical and material composition of batteries determines their size, format, and overall performance. Therefore, each battery has a different composition. However, most batteries have some common components, although their material composition may vary. The main parts of most batteries are:
- Cathode
- Anode
- Electrolyte
- Separator
- Current collectors
An electric battery is essentially a source of DC electrical energy. It converts stored chemical energy into electrical energy through an electrochemical process. This then provides a source of electromotive force to enable currents to flow in electric and electronic circuits. A typical battery consists of one or more voltaic cells.
The fundamental principle in an electrochemical cell is spontaneous redox reactions in two electrodes separated by an electrolyte, which is a substance that is ionic conductive and electrically insulated.
Chemical energy can be stored, for example, in Zn or Li, which are high-energy metals because they are not stabilized by d-electron bonding, unlike transition metals. Even though many types of batteries exist with different combinations of materials, all of them use the same principle of the oxidation-reduction reaction. Batteries are designed so that the energetically favorable redox reaction can occur only when electrons move through the external part of the circuit.
Batteries are made of an extensive range of materials resulting in different capabilities and behaviors in the functionality of the battery. The most common ones are lead, nickel, zinc, and lithium, each of them with different outputs and specific for some different purposes depending on the requirements.
Composition of Battery
Many types of electrochemical cells have been produced, with varying chemical processes and designs, including galvanic cells, electrolytic cells, fuel cells, flow cells, and voltaic piles.
- A wet cell battery has a liquid electrolyte. Other names are flooded cell since the liquid covers all internal parts, or vented cell since gases produced during operation can escape to the air. Wet cells were a precursor to dry cells and are commonly used as a learning tool for electrochemistry.
- A dry cell uses a paste electrolyte with only enough moisture to allow current to flow. Unlike a wet cell, a dry cell can operate in any orientation without spilling, as it contains no free liquid, making it suitable for portable equipment.
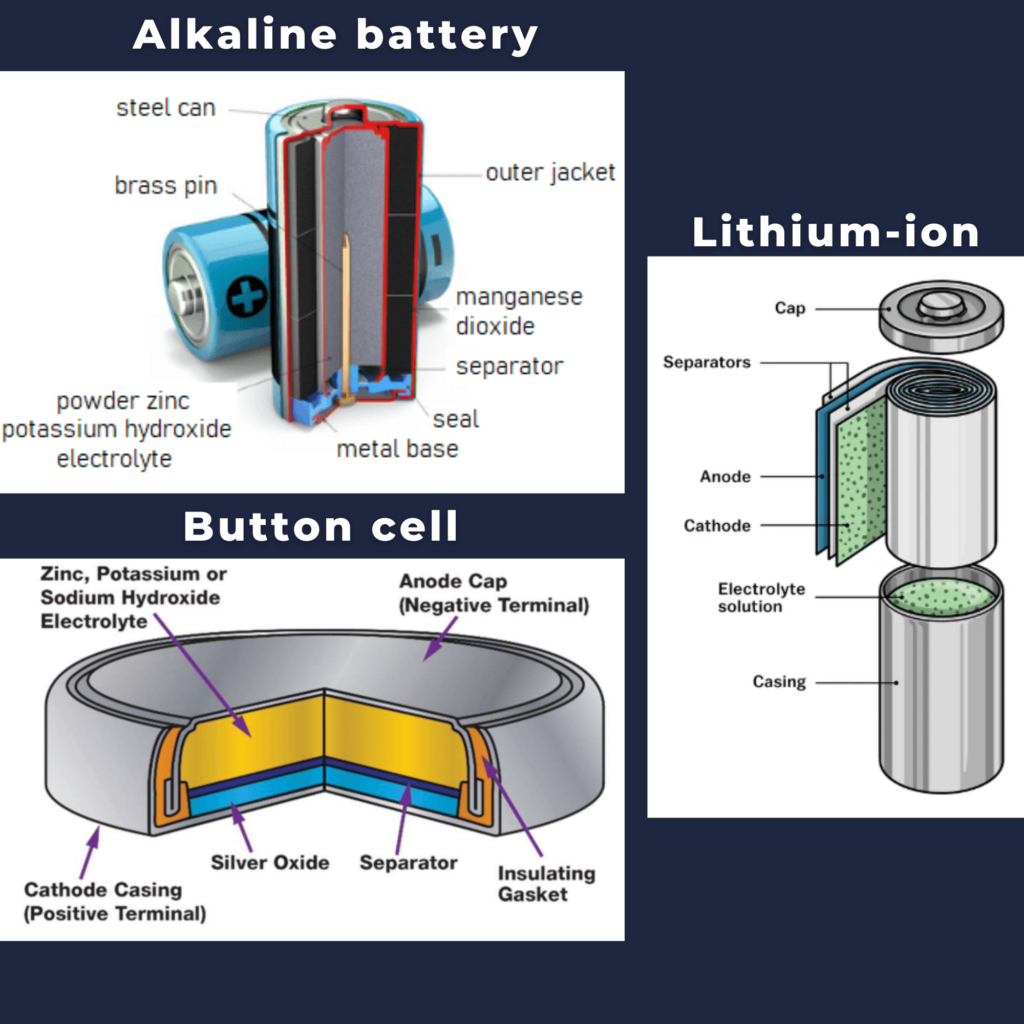
The chemical and material composition of batteries determines their size, format, and overall performance. Therefore, each battery has a different composition. However, most batteries have some common components, although their material composition may vary.
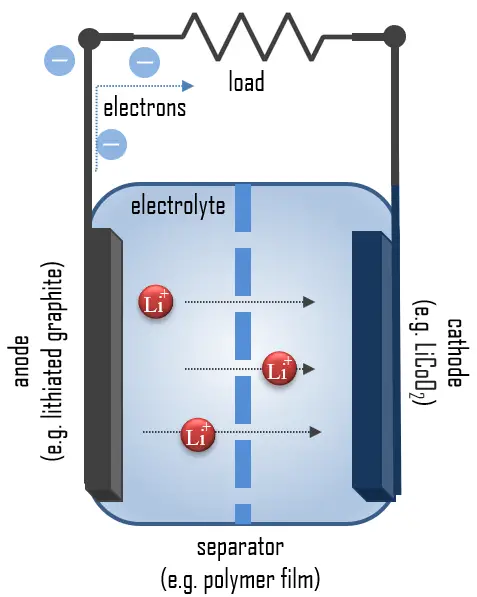
- Cathode. The cathode is the positive or oxidizing electrode that acquires electrons from the external circuit and is reduced during the electrochemical reaction. In the case of lithium batteries, cathode materials are generally constructed from LiCoO2 or LiMn2O4. For the cathode, it is important to hold a large amount of lithium without significant change in structure, have a good chemical and electrochemical stability with electrolyte, be a good electrical conductor and diffuser of lithium ions, and be of low cost.
- Anode. The anode is the negative or reducing electrode that releases electrons to the external circuit and oxidizes during an electrochemical reaction. One of the most common anode materials used today is lithiated graphite, LixC6, which is composed of graphite sheets intercalated with lithium. New materials such as those based on Silicon and other elemental blends are being researched. Lithiated graphite has a unit cell with an HCP structure.
- Electrolyte. An electrolyte is a medium containing ions that are electrically conducting through the movement of those ions but not conducting electrons. The battery electrolyte is a solution inside batteries. Depending on the type of battery, it can be a liquid or paste-like substance. The choice of electrolyte in all batteries is critical for performance as well as safety. In lithium-ion batteries, the electrolyte is typically a lithium salt dissolved in organic solvents. A good electrolyte must have low reactivity with other cell components, high ionic conductivity, low toxicity, a large window of electrochemical voltage stability (0-5V), and be thermally stable. Aqueous potassium hydroxide is employed as the electrolyte in alkaline batteries based on nickel-cadmium, nickel-hydrogen, and manganese dioxide-zinc.
- Separator. A separator is a permeable membrane placed between a battery’s anode and cathode. The main function of a separator is to keep the two electrodes apart to prevent electrical short circuits while also allowing the transport of ionic charge carriers that are needed to close the circuit during the passage of current in an electrochemical cell. Commercially available liquid electrolyte cells use microporous polyolefin materials, such as polyethylene (PE) or polypropylene (PP). Separators in Li-ions have to be electrochemically and chemically stable relative to the electrolyte and electrode materials. Functional separators that use MOF-coated membranes to perform the dual functions of the electrolyte and separator are being developed to support the design of high-performance Li-metal batteries for high-energy systems in electric vehicles and electric aircraft.
- Current Collectors. Current collectors comprise the battery component responsible for transferring the flow of electrons from the electrodes to an external circuit. Several types of current collectors are available: mesh, foam, and foil. To minimize overall size and improve the volumetric capacity of cells, metallic foils which are thin and light are preferred. Current collectors are an electrochemically inactive volume in the cell but form the substrate that the electrochemically active materials are applied. Active materials are applied onto the thin current collectors with a conducting agent and an adhesive binder. Hence, current collectors should possess high electrical conductivity to reduce cell resistance as well as chemical stability in contact with liquid electrolyte over the operating voltage window of electrodes.
The alkaline battery consists of five parts:
- Inner current collector (pin)
- Anode. The active material in the anode is Zn. With a standard electrode potential (SEP) of −0.76 volts, zinc is used as an anode material for batteries. (More reactive lithium (SEP −3.04 V) is used for anodes in lithium batteries ).
- Separator. A separator is a permeable membrane placed between a battery’s anode and cathode. For example, non-woven, fibrous fabric that separates the electrodes.
- Cathode. The active material of the cathode is manganese dioxide. The principal use for MnO2 is for dry-cell batteries, such as the alkaline battery and the zinc-carbon battery.
- Electrolyte. Aqueous potassium hydroxide is employed as the electrolyte in alkaline batteries.
- Outer current collector (can)
[alkaline-battery]
Characteristics of Electric Batteries
To compare and understand the capability of each battery, some important parameters are characteristic of each battery, also within a type of battery. These parameters are a reference when a battery is needed, and specific qualities are required since batteries are used in all types of devices and for infinite purposes.
Cell Voltage
The voltage of electric batteries is created by the potential difference of the materials that compose the positive and negative electrodes in the electrochemical reaction.
Cut-off Voltage
The cut-off voltage is the minimum allowable voltage. It is this voltage that generally defines the “empty” state of the battery.
Capacity
The coulometric capacity is the total Amp-hours available when the battery is discharged at a certain discharge current from 100% SOC to the cut-off voltage.
C-rate of Battery
The cut-off voltage is the minimum allowable voltage. It is this voltage that generally defines the “empty” state of the battery.
Self-discharge
Batteries gradually self-discharge even if not connected and delivering current. This is due to non-current-producing “side” chemical reactions that occur within the cell even when no load is applied.
Degradation
Some degradation of rechargeable batteries occurs on each charge–discharge cycle. Degradation usually occurs because electrolyte migrates away from the electrodes or because active material detaches from the electrodes.
Depth of Discharge
Depth of discharge is a measure of how much energy has been withdrawn from a battery and is expressed as a percentage of full capacity. For example, a 100 Ah battery from which 40 Ah has been withdrawn has undergone a 40% depth of discharge (DOD).
State of Charge
The state of charge refers to the amount of charge in a battery relative to its predefined “full” and “empty” states i.e. the amount of charge in Amp-hours left in the battery.
Frequently asked questions
A typical battery needs 4 parts to create electricity: Anode – the negative or reducing electrode that releases electrons to the external circuit. Cathode – the positive or oxidizing electrode that acquires electrons from the external circuit. Electrolyte – a chemical paste that separates the anode and cathode and transforms chemical energy into electrical energy.
In practice, the alkaline batteries and the rechargeable batteries can be used interchangeably in sets. They have only different voltage characteristics. It is given by their different chemistry. Primary cells gradually drop in voltage from use, they start at 1.5 volts, drop to 1.2 and continue to 1.0 where the appliance stops working. Secondary cells operate more uniformly even with only 1.2 volts, they have flat discharge where they pretty much stay at 1.2 volts until depleted and then drop off very quickly to below 1.0 volts.
A lithium-ion polymer (LiPo) battery (also known as Li-pol, lithium-poly, and other names) is a type of Li-ion battery with a polymer electrolyte instead of a liquid electrolyte. All use a high conductivity gel polymer as the electrolyte. Lithium polymer cells have evolved from lithium-ion and lithium-metal batteries.









