30-second summary
Elementary Electric Charge
The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge −1 e. This elementary charge is a fundamental physical constant.
e = 1.602176487 x 10-19C
One coulomb represents the negative of the total charge of about 6 x 1018 electrons.

About Electric Charge
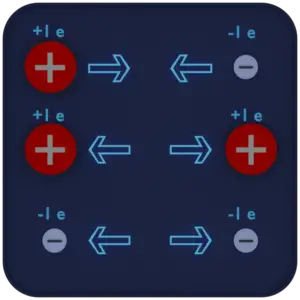
An electric charge is a physical quantity and property of matter that causes it to experience a force when placed in an electromagnetic field. There are two types of electric charge: positive, transmitted by protons, and negative, transmitted by electrons. If the total charge is zero, it is said to be neutral. The same charges are repelled and the opposite charges are attracted. These facts are known as the First Law of Electrostatics and are sometimes referred to as the law of electrical charges.
Elementary Charge
The most fundamental unit of charge is the magnitude of the charge of an electron or a proton, which is denoted by e. The most precise value available is:
e = 1.602176487 x 10-19C
One coulomb represents the negative of the total charge of about 6 x 1018 electrons.
We rarely encounter charges as large as a coulomb. Charges produced by rubbing ordinary objects (such as a comb or plastic ruler) are typically around a microcoulomb (?C = 10-6 C) or less.
The proton has charge + e and electron – e. The charge is quantized; it comes in integer multiples of individual small units called the elementary charge, e, which is the smallest charge which can exist freely (particles called quarks have smaller charges, multiples of ⅓ e, but they are only found in combination, and always combine to form particles with integer charge). The proton has a quark composition of uud, and so its charge quantum number is:
q(uud) = 2/3 + 2/3 + (-1/3) = +1e.
The neutron has a quark composition of udd, and its charge quantum number is therefore:
q(udd) = 2/3 + (-1/3) + (-1/3) = 0
Since the neutron has no net electric charge, it is not affected by electric forces, but the neutron does have a slight distribution of electric charge within it. This is caused by by its internal quark structure. This results in non-zero magnetic moment (dipole moment) of the neutron. Therefore the neutron interacts also via electromagnetic interaction, but much weaker than the proton.
Electric Charge of Antiparticles
Theoretically, a particle and its anti-particle (for example, a proton and an antiproton) have the same mass, but opposite electric charge, and other differences in quantum numbers. For example, for every quark there is a corresponding type of antiparticle. The antiquarks have the same mass, mean lifetime, and spin as their respective quarks, but the electric charge and other charges have the opposite sign. That means a proton has positive charge while an antiproton has negative charge and therefore they attract each other. The antiparticle of the electron is called the positron; it is identical to the electron except that it carries electrical and other charges of the opposite sign. When an electron collides with a positron, both particles can be totally annihilated, producing gamma ray photons.

