Barium titanate is a ferroelectric material with unique properties, used in capacitors, sensors, actuators, transducers, electro-optic devices, and energy storage.
Barium Titanate as Ferroelectric Material
Introduction
Barium titanate (BaTiO3) is a versatile and widely studied ferroelectric material that has attracted significant attention due to its fascinating properties and numerous applications. Ferroelectric materials are characterized by their ability to exhibit spontaneous electric polarization that can be reversed by an applied electric field. This characteristic makes them ideal for use in various industries, including electronics, sensors, and energy storage devices. In this article, we will discuss the unique properties of barium titanate, its synthesis methods, and its applications in modern technology.
Unique Properties of Barium Titanate
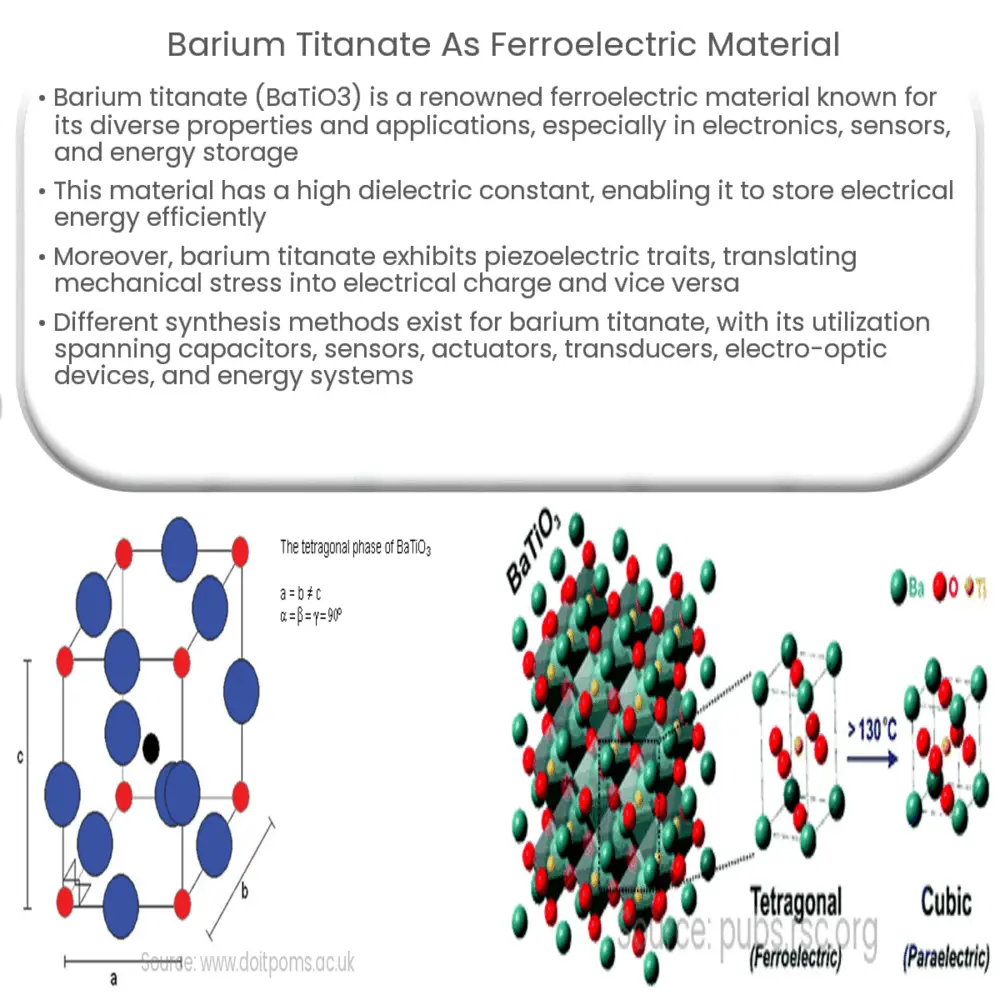
Barium titanate is a perovskite-type compound with a chemical formula of BaTiO3. It is a ferroelectric material with a high dielectric constant, which means it has a high ability to store electrical energy. Barium titanate undergoes a series of phase transitions depending on the temperature, which leads to changes in its crystal structure and ultimately its dielectric properties. The most significant phase transition occurs at the Curie temperature, where the material transforms from a non-ferroelectric (paraelectric) phase to a ferroelectric phase.
In addition to its high dielectric constant, barium titanate also exhibits piezoelectric properties, meaning it can convert mechanical stress into electrical charge and vice versa. This characteristic makes barium titanate an essential material for applications such as sensors, actuators, and transducers. Furthermore, its optical properties, including high refractive index and transparency in the visible range, make it suitable for use in electro-optic devices.
Synthesis Methods of Barium Titanate
There are several methods available for synthesizing barium titanate, each with its advantages and drawbacks. Some of the most common synthesis techniques include solid-state reaction, sol-gel, hydrothermal, and chemical vapor deposition (CVD). The choice of synthesis method often depends on the desired properties, purity, and application of the final product.
The solid-state reaction is the most traditional method for preparing barium titanate, involving the reaction of barium carbonate (BaCO3) and titanium dioxide (TiO2) at high temperatures. This technique is relatively simple and cost-effective but may result in impurities and a limited control over the particle size and morphology.
The sol-gel method involves the formation of a colloidal suspension, followed by gelation, drying, and heat treatment to produce the desired barium titanate material. This technique allows for better control over the particle size and morphology, resulting in a more uniform product. The hydrothermal method involves the synthesis of barium titanate under high-pressure and high-temperature aqueous conditions, providing excellent control over the crystal structure and particle size.
Lastly, the chemical vapor deposition technique involves the deposition of barium and titanium precursors onto a substrate, followed by a reaction to form barium titanate. This method is particularly useful for producing thin films and coatings with excellent uniformity and purity.
Applications of Barium Titanate
Barium titanate’s unique combination of ferroelectric, piezoelectric, and optical properties has led to its widespread use in various applications across different industries. Some of the most prominent applications include:
1. Capacitors
The high dielectric constant of barium titanate makes it a suitable material for use in multilayer ceramic capacitors (MLCCs), which are essential components in electronic devices such as computers, smartphones, and televisions. The high capacitance offered by barium titanate-based MLCCs enables the miniaturization of electronic devices while maintaining excellent performance.
2. Sensors and Actuators
Barium titanate’s piezoelectric properties make it ideal for applications such as sensors and actuators. For example, barium titanate-based sensors can detect pressure, force, or acceleration changes and convert them into electrical signals, while barium titanate-based actuators can convert electrical signals into mechanical motion. These applications can be found in automotive systems, medical devices, and industrial machinery.
3. Transducers
The piezoelectric properties of barium titanate are also useful in the production of ultrasonic transducers, which can convert electrical signals into ultrasonic waves and vice versa. Ultrasonic transducers are widely used in medical imaging, non-destructive testing, and underwater communication systems.
4. Electro-optic Devices
Due to its high refractive index and transparency in the visible range, barium titanate is employed in electro-optic devices such as modulators, switches, and waveguides. These devices are essential components in optical communication systems, allowing for high-speed data transmission with minimal loss.
5. Energy Storage and Harvesting
Barium titanate’s high dielectric constant and energy density make it a promising material for energy storage applications, such as in supercapacitors and high-energy-density capacitors. Additionally, its piezoelectric properties can be harnessed for energy harvesting applications, where ambient mechanical vibrations can be converted into electrical energy to power small electronic devices.
Conclusion
In summary, barium titanate is a versatile ferroelectric material that has found widespread use across various industries due to its unique combination of properties. With continued research and development, barium titanate is expected to play an even more significant role in the advancement of technologies such as electronics, sensors, actuators, energy storage, and optical communication systems.

